
Hypoxia in shoot apical meristem
Most plants suffer during low oxygen conditions like e.g. flood events, but now researchers from the University of Copenhagen, University of Pisa, RWTH Aachen University and University of Heidelberg have discovered that the production of new leaves in plants is dependent on hypoxic conditions.
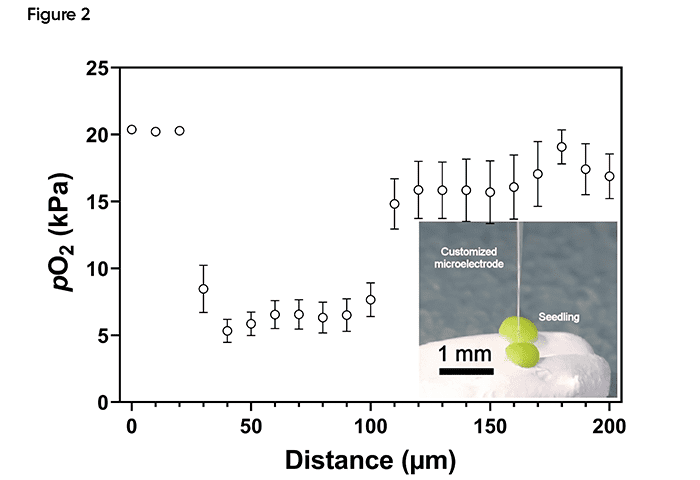
The researchers used a customized Unisense oxygen microsensor with a diameter of only 3 µm to measure the oxygen level inside the shoot apical meristem of plants (Figure 1). Microprofiles with a step size of 10 µm were made throughout the tissue using the MicroProfiling System.

The results showed a low oxygen level inside the tissue area consisting of only approximately 30 cells (Figure 2).
The plants maintained a low oxygen level in the shoot apical meristem and treatment with high oxygen concentrations stopped the development of the stem cells.
The study revealed that hypoxia is a prerequisite for the function of stem cells in plants which is a phenomenon also seen in human stem cells.
Publication: Weits et al. An apical niche sets the pace of shoot meristem activity, Nature, 569, pages714–717 (2019).
You can read the full article here: http://dx.doi.org/10.1038/s41586-019-1203-6
Related Publications
Related Products
High performance oxygen microsensor

UniAmp Multi Channel for all Unisense sensors and electrodes including optical sensors
Microprofiles with extreme accuracy, high spatial and temporal resolution

Water resistant Field MicroProfiling System to generate, study, and analyze microprofiles

Optical oxygen sensor technology
Detect hydrogen sulfide in your sample

Economic amplifier portfolio for single analytes - O2, pH/mV, H2, N2O or H2S
Measure dissolved and gaseous hydrogen with high-precision

Optode meter for accurate quantification

Measure dissolved and gaseous nitrous oxide
Measure minute concentrations of nitric oxide
Miniaturized pH electrode
Measure redox potential in microenvironments
Microelectrode to measure electric potential

Reference electrode for pH or Redox microelectrodes
